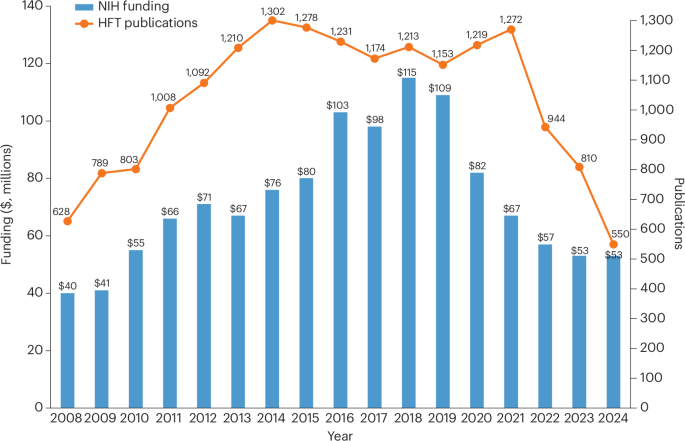
Human fetal tissue (HFT) remains an essential biomedical research tool that enables crucial advances in vaccine production, developmental biology and disease modeling1. Despite the utility of HFT and the lack of suitable alternatives, its use in research remains contentious, regularly sparking ethical controversy and political debate2. In 2019, the first Trump administratifincluding the requirementon imposed restrictions on how the US National Institutes of Health (NIH) funds HFT research, which the Biden administration then partially rescinded in 2021. Recently, in January 2026, the second Trump administration announced a complete and immediate ban on NIH funding of research using HFT from elective abortions, justifying the policy based on a “sharp decline” in NIH-supported HFT research since 2019 and a desire to prioritize NIH resources toward “biomedical research models with more relevance to today’s rapidly evolving research ecosystem”3. To better contextualize the decline in HFT research funding cited as justification for this new ban, we assessed how the 2019 restrictions and 2021 partial repeal impacted the HFT research enterprise.
The 2019 restrictions took several forms, including creating an Ethics Advisory Board (EAB) to review HFT proposals recommended for funding through NIH’s peer review process, requiring additional justification for HFT use within each proposal, and banning HFT use in intramural research and research supported by certain training grants and fellowships2. In 2021, the Biden administration rescinded certain elements of these restrictions, including the requirement for EAB review and the prohibition on HFT usage in intramural research2. Other restrictions, including the requirement for additional HFT justification and the ban on using HFT in training grants and fellowships, remained in place throughout the Biden administration and into the second Trump administration.
— Source: Nature Medicine (https://www.nature.com/articles/s41591-026-04268-y)